Universities
Universities and research institutes, and other academic institutions play an important role in improving the reporting of animal research by promoting the ARRIVE guidelines.
As important centres of scientific advancement and developers of future research leaders, universities are ideally placed to raise awareness of the standards expected when reporting animal research. Encourage research staff and students to use the ARRIVE guidelines in the planning, conduct and reporting of all animal experiments.
How can universities use and promote the ARRIVE guidelines?
- Institutional policies on animal research: Encourage researchers to publish in line with the ARRIVE guidelines. In the UK, the major funders of bioscience research (e.g. the Wellcome Trust, MRC and BBSRC) have endorsed the ARRIVE guidelines; compliance with the guidelines is a condition of grant funding. In the US, the NIH includes the ARRIVE guidelines in their Principles and Guidelines for Reporting Preclinical Research.
- Public statement: Show support for the ARRIVE guidelines through an online public statement, to be included in codes for research ethics, policy on animal use in scientific procedures and internal communications. For example:
|
The University of Nottingham fully supports and endorses the ARRIVE guidelines developed by the NC3Rs, to improve the design and reporting of studies involving animals. The ARRIVE guidelines are actively promoted by the AWERB for use by licence applicants; during training courses and within research group seminars. University of Nottingham - Policy on the Use of Animals in Research |
- IACUC or licensee training courses: Provide information about the ARRIVE guidelines during training courses for IACUC certification, or project and personal licencees. Local ethical review committees such as the AWERB, the IACUC or their equivalent also provide a useful platform for dissemination of the guidelines and associated resources across the institution.
- Research training courses: Include information about the ARRIVE guidelines on research skills courses for Masters students, PhD students or early career researchers, for example courses on experimental design, research statistics or writing for publications. Teaching resources can be found on our specific training resources pages.
- Ethical review: Request information in line with the ARRIVE guidelines as part of ethical review applications, and encourage the use of the guidelines during experimental design and project planning:
|
Refer to the ARRIVE guidelines and ARRIVE guidelines checklist as an aid to project planning and experimental design considerations. NOTE - Animal Research Ethics Board (AREB) review of an Animal Use Protocol will consider ARRIVE guidelines checklist items and recommendations as appropriate. University of Saskatchewan - Animal Research Ethics Board |
To endorse the ARRIVE guidelines, share suggestions on the use of ARRIVE in universities and research institutes, or suggest the development of new resources, please email arrive@nc3rs.org.uk.
Institutions endorsing the ARRIVE guidelines 2.0
Here you can find academic institutions who endorse the ARRIVE guidelines. The map below illustrates those countries (or international organisations) in which endorsing institutions are located, and a list can be found below.
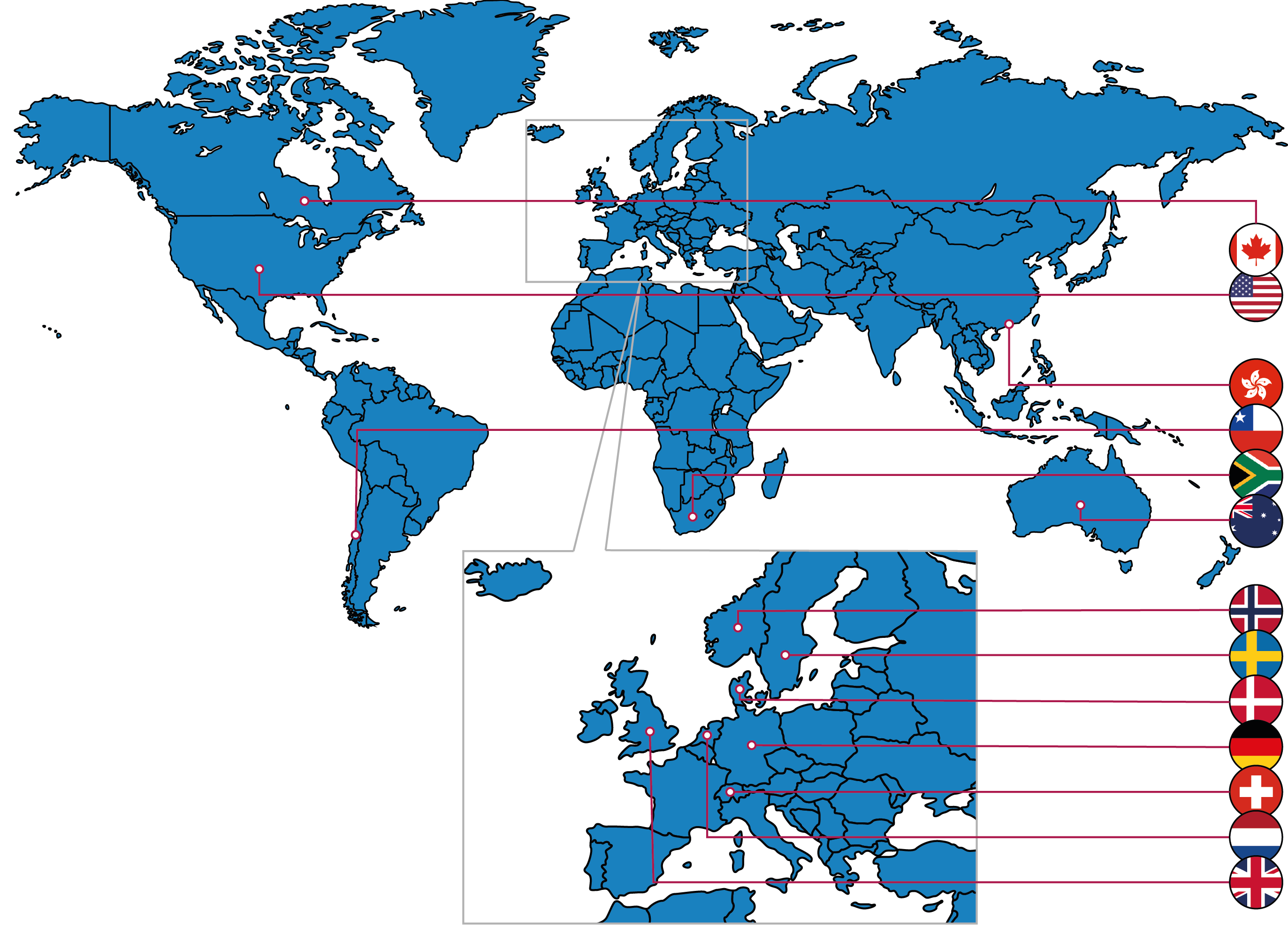 |
|



